
Vaping giant Juul pushes for more addictive e‑cigarettes
The vaping giant Juul is lobbying for stronger and more addictive e-cigarettes to be sold in the UK after Brexit, despite being blamed by many for a teen vaping “epidemic” in the US.
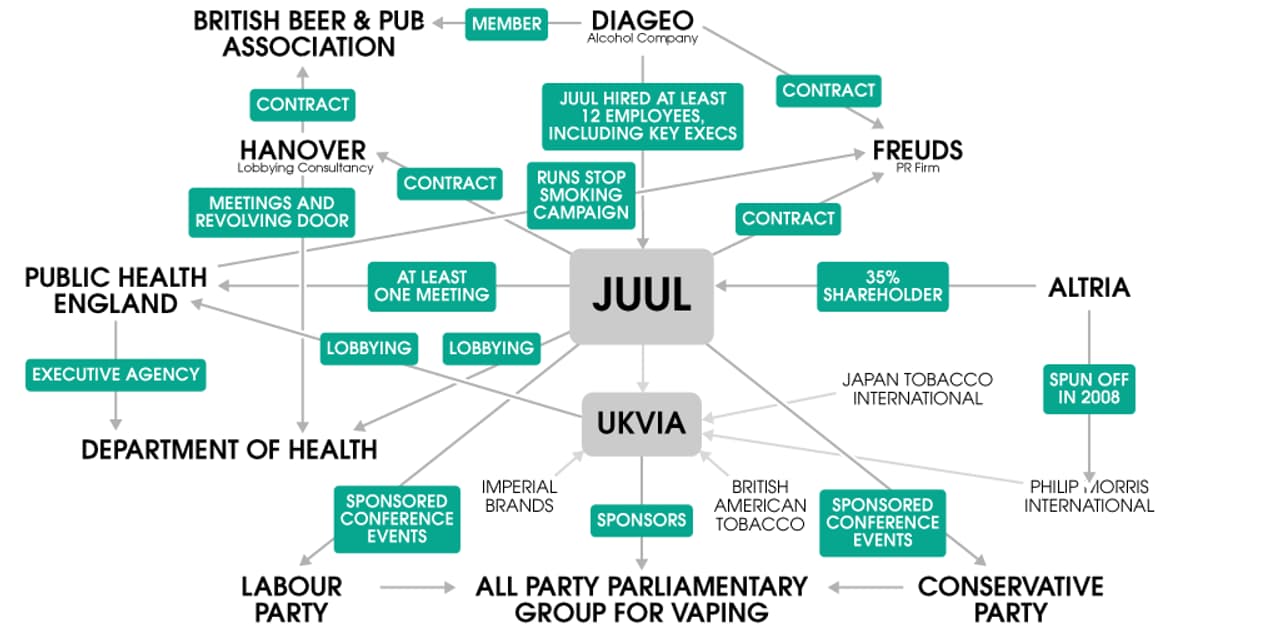
The company has worked behind the scenes to build a substantial PR and lobbying operation across Westminster, including hiring executives with ties to government, an investigation by the Bureau has found. It is also working to lift stringent advertising restrictions on its products.
Juul’s public affairs playbook appears to mirror that of the drinks industry, which has faced years of criticism over fostering a binge-drinking culture among young people. Juul has hired the same PR agency used by Diageo – which owns Smirnoff and Guinness – and employs several of its former staff.
In a submission to the Department of Health last year obtained by the Bureau under Freedom of Information rules, Juul said that Brexit offered an opportunity to relax advertising restrictions and review the “arbitrary limit” on the strength of nicotine in e-cigarettes set by the European Union.
Juul joined the UK Vaping Industry Association (UKVIA) earlier this year, a trade group which is also calling on the government to strip back strict EU laws. The Department of Health has pledged to “identify areas where we can sensibly deregulate”.
The company is facing a huge backlash in the US, its home market, where more than a quarter of high school students vape, and is embarking on an ambitious global expansion plan, partly funded by an $12bn investment from Altria, the maker of Marlboro cigarettes and Juul’s largest shareholder.
The Bureau has tracked Juul’s lobbying in countries all over the world, revealing the UK as a key market, where the company has seen “rapid growth” since its launch last year.
Some of the fruit and dessert-based flavours on sale in the UK have been pulled from US shelves after criticism that they appealed to children. Juul’s UK products contain much less nicotine than its US pods – which are strong even for that market – because regulations set the limit on nicotine content at 20 mg/ml.
Juul and UKVIA say the government should lift this “arbitrary” cap after Brexit. Martin McKee, professor of European public health at the London School of Hygiene and Tropical Medicine, said: “It’s insane. Given everything that’s going on at the moment, it’s mad.
“The trick is to get people addicted to nicotine and that’s going to be easier with higher concentrations so it’s not surprising that [the industry] wants to do that ... But why would we want to when we’ve seen what has happened in the US?”
UKVIA said: “Allowing for a range of nicotine strengths provides heavy smokers with the tools they need to make the switch away from cigarettes, which are far more harmful than vaping.”
Pushing the limits
The government has already committed to a post-Brexit review of the EU’s e-cigarette regulations. It will “reappraise” these restrictions, which in effect ban advertising on TV, radio, in print and online.

UKVIA – which also represents major tobacco companies, including Philip Morris International and British American Tobacco – says EU laws “severely curtail the ability of the industry to communicate the public health potential of vaping to smokers”.
Some restrictions have already been lifted. UKVIA and tobacco companies – along with Public Health England (PHE) and a handful of other health groups – supported the Advertising Standards Agency when it relaxed guidelines last year, so that health claims made in marketing materials no longer require authorisation by the UK’s Medicines and Healthcare products Regulatory Agency.
Professor Dame Parveen Kumar, of the British Medical Association, opposed the changes and said any further weakening of ad rules needed “serious thought” about the potential impact on public health.
Professor Jonathan Grigg, a paediatrician and leading expert on the effects of air pollution, echoed her concerns, saying the devices “should not be visible to young people in any way”. He added: “There’s absolutely no reason why these devices can’t be behind the shutters that we see for cigarettes until we absolutely know what the long-term effects of these devices are.”
Public Health England says it has a “balanced policy”. As vaping is significantly safer than smoking, it believes any barriers to smokers accessing e-cigarettes should be kept to a minimum. But critics are calling for it to re-evaluate its laissez-faire stance and raising questions over its seemingly positive attitude towards the vaping industry.
The criticism intensified after dozens of deaths in the US caused by vaping-related respiratory illnesses. The majority of cases are believed to have been caused by black market products, but some have blamed Juul for the dramatic rise in vaping among teens generally, after the company ran a controversial ad campaign centred on young models and social media influencers.
 Juul's US advertising campaign was criticised for appealing to a young audience
Juul's US advertising campaign was criticised for appealing to a young audience
 Their UK adverts have a much more sombre tone
Their UK adverts have a much more sombre tone
UKVIA phoned PHE to ask if they were going to make a statement on the US deaths. In an email to an official on PHE’s tobacco control team in August, UKVIA said: “It was good to speak to you earlier, and thanks for your information that PHE’s are [sic] planning to put out a statement.” It added: “UKVIA is very happy to engage with PHE to provide any advice or guidance and share best practice too.”
PHE’s statement said “a full investigation is not yet available” but added that “unlike the US, all e-cigarette products in the UK are tightly regulated for quality and safety”.
A month later Martin Dockrell, the head of tobacco control at PHE, was interviewed on BBC Breakfast and told viewers that none of the deaths in the US were linked to products sold in the UK. Later that week, a mother in the US filed a wrongful death lawsuit against Juul. She believes Juul is responsible for the death of her teenage son, who was a heavy user and suffered from breathing difficulties. The case has not yet been heard. PHE said Juul’s higher nicotine pods are not available in the UK.
The Bureau understands that PHE has consulted the US Centers for Disease Control and Prevention about the death of a Briton believed to have been linked to vaping.
Martin McKee said: “Public Health England keeps saying [vaping] it’s not a problem even when there’s plenty of evidence it is a problem ... It’s dug itself into a hole and it can’t get out.”
 Martin Dockrell
BBC
Martin Dockrell
BBC
PHE told the Bureau: “Smoking kills around 220 people a day. PHE’s independent e-cigarette reviews are world leading and the evidence is clear that, while not without risks, vaping is a fraction of the risk of smoking and an effective way to quit. But we are also clear, if you’ve never been a smoker, don’t vape."
Juul said in a statement: “We go above and beyond to combat the issue of youth access, appeal and use of vapour products, while at the same time ensuring that adult smokers maintain access to a product that is helping millions of them switch from combustible cigarettes.”
Schmoozing and boozing
After an onslaught of federal and congressional investigations in the US, Juul promised to stop lobbying in Washington – but in the UK, where no such promises have been made, it has been doing just the opposite.
In May, Juul hired Max Chambers, David Cameron’s former speechwriter, as its UK director of government and public affairs. Prior to joining Juul, Chambers worked for Public.io, a company set up by one of his former Downing Street colleagues that works to help secure public sector contracts for its tech start-up clients. The job kept Chambers in contact with senior members of government, with ministerial disclosures showing he met with the Cabinet Office and the Home Office early this year. Juul said Chambers had “no involvement in the formulation of e-cigarette regulation in the UK” and had no involvement with Juul while he was in Downing Street.
As near-daily reports about Juul’s role in a teen vaping “epidemic” hit the press in the US in September, Chambers was on the party conference circuit, with Juul sponsoring panels at Labour and Conservative events. At the Tory party conference, Juul held two closed events for MPs and their special advisers. It also sponsored health panels at a non-partisan political conference in August, with the junior health minister Nicola Blackwood as one of the speakers.
In September last year, James Kent, then the special adviser for health to the prime minister Theresa May, disclosed that he’d received “USB chargers” as a gift from Juul. The Bureau has learnt that Juul met with Kent to discuss how e-cigarettes could help adult smokers “move away from combustible cigarettes”.

To build its lobbying team, Juul has turned to two agencies with clout – Hanover Communications and Freuds. Both have also been employed by the drinks industry; Juul has hired at least 10 former Diageo employees to work in its London office, including its director of finance, and appears to be emulating Diageo’s approach to lobbying for self-regulation.
The British Beer and Pub Association, of which Diageo is a member, works with Hanover and launched ‘Challenge 21’ in response to growing calls to stop underage sales. A self-regulated age-verification scheme in pubs and retailers, it was seen by some as an attempt to stave off stricter legislation.
Both Juul and UKVIA have endorsed a similar scheme for e-cigarettes.
Hanover enjoys strong connections to government, particularly the Department of Health and its agencies, having worked in the past for both PHE and the NHS. The company’s chief executive was once Sir John Major’s press secretary, and Hanover’s head of health, Andrew Harrison, worked at the department in the 1990s with Simon Stevens, now the head of NHS England. Hanover said it has no “ties” with public health agencies.
In May, the health secretary Matt Hancock attended a breakfast hosted by Hanover. A few months later, Emma Dean left her job at Hanover to become a special adviser at the Department of Health.
Freuds, meanwhile, has faced conflict of interest concerns after the agency was brought in to launch PHE’s stop smoking campaign. Freuds said it stopped working for Juul earlier this year and it “will never work for a tobacco company”. However, Juul’s largest shareholder is tobacco giant Altria, which sells Marlboro cigarettes in the US.
The executive who led the Public Health England and Diageo accounts for Freuds was hired by Juul as director of marketing communications last year, according to LinkedIn.
Until recently, Juul was conducting a “confidential search for a chief medical officer”, according to an email sent to one of the UK’s leading e-cigarette experts seen by the Bureau. The chief medical officer would report to Juul’s CEO and have a “significant impact on the future of the organisation”, the recruiter said. As well as someone who could oversee Juul’s research, they would also be responsible for “partnership with the medical and research communities, regulatory stakeholders, and industry”.
In July, Politico reported Juul had hired an expert in the effects of nicotine on the teenage brain as its executive medical officer in the US. The move was criticised by some as mimicking a Big Tobacco tactic to create credibility and buy influence.
Juul told the Bureau it is no longer recruiting for the position of chief medical officer.
Beyond Westminster
Juul’s lobbying efforts extend beyond the Westminster bubble. The company has been careful with its public image in the UK, making clear in its advertisements that it is only intended for adult smokers trying to quit. But the Bureau’s findings suggest that behind closed doors the company has been eager to seek to influence strict EU regulations.
PHE met with Juul employees in April last year, before its UK launch, to discuss EU legislation. While PHE is required to disclose the meeting, official records state “the discussion related to regulatory issues and was commercial in confidence”.
PHE told the Bureau: “We are ranked top in the world for resisting interference from the tobacco industry with PHE cited specifically for our transparency.”
During another private meeting with EU tax officials in March, Juul argued any tax on e-cigarettes should be based on the volume of the liquid, not on the strength of nicotine. While the EU does not require member states to place any additional tax on e-cigarettes – as it does for regular cigarettes – it is reviewing whether it should. Britain does not put a specific tax on e-cigarettes, but about half of EU member states do.
Juul’s proposed rules, which lift the limit on nicotine content, would pave the way for it to sell its stronger pods in Europe without any increase in price.
Matt Myers, president of Campaign for Tobacco-Free Kids, which has closely monitored Juul’s rise in the US and its emergence as a global business, said: “Juul is following the worst of what the tobacco industry does, hiring high paid, well-connected PR and lobbying firms to overcome the lack of science, the wrongful behaviour and public health concerns.
“It is hard to miss the irony that Juul and its defenders point to the United Kingdom, where it claims smoking cessation is up and no youth crisis exists, even as it is fighting to undermine the very laws and regulations that have prevented the youth epidemic in the UK.”
Illustration by Rebecca Hendin
Our reporting on tobacco is part of our Global Health project which has a number of funders. Smoke Screen is funded by Vital Strategies. None of our funders have any influence over the Bureau’s editorial decisions or output.





